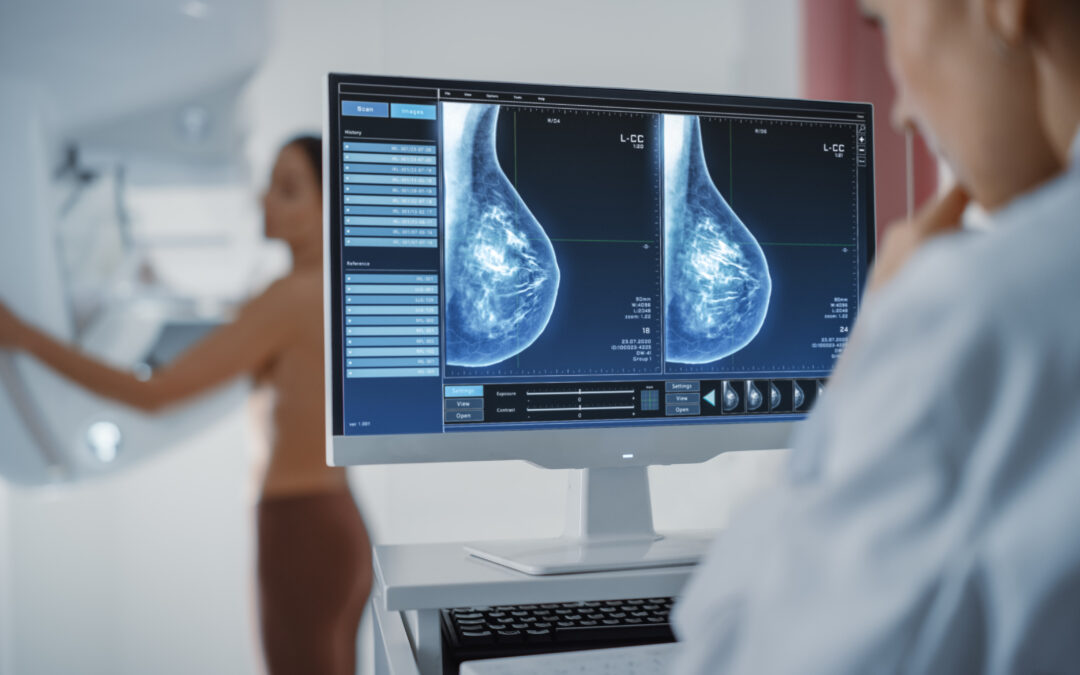
Exact Sciences (NASDAQ: EXAS) Presents Data Demonstrating Advancement in Blood-based Colorectal Cancer Screening at ESMO 2024.
Exact Sciences Corp. (NASDAQ: EXAS), a leading provider of cancer screening and diagnostic tests, today announced performance data for its blood-based colorectal cancer (CRC) screening test. Results show sensitivities of 88.3% for CRC and 31.2% for advanced precancerous lesions at specificity of 90.1% for negative samples confirmed by colonoscopy. Results were presented today at the European Society for Medical Oncology (ESMO) Congress in an oral presentation titled, “Organ-specific performance of a multi-analyte, multi-cancer early detection (MCED) blood test in a prospectively collected cohort.”
“The Exact Sciences team is constantly innovating to help close the screening gap,” said Kevin Conroy, chairman and CEO, Exact Sciences. “The insights that led to this innovation reflect our understanding of the biology of cancer and the power of our scientific capabilities. We took a unique scientific approach to developing this test by combining a novel panel of markers. This led to data that improve upon what we previously thought was possible with a blood-based colorectal cancer screening test.”
To optimize the final test algorithm, Exact Sciences designed a study to simulate the screening population in the United States and better predict real-world, prospective performance of a novel test. The study consisted of more than 3,000 blood samples, including approximately 2,900 blinded, prospectively collected samples from the pivotal BLUE-C study. This analysis was prespecified with the U.S. Food and Drug Administration (FDA) and the samples will be excluded from the final clinical validation. The study also included more than 90 advanced precancerous lesions, the majority of which were prospectively collected, and 60 case-collected colorectal cancer samples. In the pivotal BLUE-C study results, performance degradation is expected for advanced precancerous lesion sensitivity and overall CRC sensitivity.
Results of this study show the potential of a novel, highly discriminate blood-based panel of methylated DNA markers and an impactful, new marker class to detect advanced precancerous lesions and cancers at an attractive cost profile. The company will implement the innovative marker class on a new testing platform and complete additional analytical studies to support an FDA submission.
“A blood-based colorectal cancer screening test that can detect advanced precancerous lesions at a level comparable to the FIT test would be a breakthrough in this field,” said Paul Limburg, MD, MPH, AGAF, chief medical officer for Screening, Exact Sciences. “Results from this large, well-designed study show progress toward that goal and move us one step closer toward providing average-risk patients with another non-invasive screening option.”
BLUE-C results for Exact Sciences’ blood-based CRC screening test are now expected in the first half of 2025. Exact Sciences plans to use these results to support an FDA submission and approval and to make the blood-based CRC screening test available broadly. If approved, the blood-based CRC screening test could provide another testing option for 60 million unscreened people1 in the United States. It would be supported by Exact Sciences’ commercial infrastructure and ExactNexus™ technology platform, making electronic ordering and resulting seamless for more than 350 health systems.
The company also presented data from its multi-cancer early detection (MCED) blood test, assessing organ-specific performance of methylation and protein biomarkers in a prospectively collected cohort of samples from its ASCEND 2 study. The analysis indicated an overall sensitivity of 54.8% with 98.5% specificity in cancers without standard-of-care screening options (excluding lung) and 63.7% in the six most aggressive cancers with the shortest survival rates (esophagus, liver, lung, ovarian, pancreatic, and stomach). These findings highlight the potential clinical value of using multiple biomarkers to detect various cancer types, including the most aggressive and those without recommended screening options.